Subject:
Coronary Computed Tomography Angiography With Selective Noninvasive Fractional Flow Reserve
Description:
_______________________________________________________________________________________
IMPORTANT NOTE:
The purpose of this policy is to provide general information applicable to the administration of health benefits that Horizon Blue Cross Blue Shield of New Jersey and Horizon Healthcare of New Jersey, Inc. (collectively “Horizon BCBSNJ”) insures or administers. If the member’s contract benefits differ from the medical policy, the contract prevails. Although a service, supply or procedure may be medically necessary, it may be subject to limitations and/or exclusions under a member’s benefit plan. If a service, supply or procedure is not covered and the member proceeds to obtain the service, supply or procedure, the member may be responsible for the cost. Decisions regarding treatment and treatment plans are the responsibility of the physician. This policy is not intended to direct the course of clinical care a physician provides to a member, and it does not replace a physician’s independent professional clinical judgment or duty to exercise special knowledge and skill in the treatment of Horizon BCBSNJ members. Horizon BCBSNJ is not responsible for, does not provide, and does not hold itself out as a provider of medical care. The physician remains responsible for the quality and type of health care services provided to a Horizon BCBSNJ member.
Horizon BCBSNJ medical policies do not constitute medical advice, authorization, certification, approval, explanation of benefits, offer of coverage, contract or guarantee of payment.
__________________________________________________________________________________________________________________________
Invasive coronary angiography (ICA) is clinically useful in stable ischemic heart disease when there is coronary artery obstruction that may benefit from revascularization. However, many individuals currently undergoing ICA will not benefit from revascularization. Therefore, if there are noninvasive alternatives to guide decisions about the use of ICA to spare individuals from unnecessary ICA, there is potential to improve health outcomes. Using noninvasive measurement of fractional flow reserve (FFR) as part of a noninvasive imaging strategy may be beneficial to avoid the need for ICA.
| Populations | Interventions | Comparators | Outcomes |
Individuals:
- With stable chest pain at intermediate risk of coronary artery disease being considered for invasive coronary angiography
| Intervention of interest are:
- Noninvasive fractional flow reserve measurement following positive coronary computed tomography angiography
| Comparator of interest are:
- Coronary computed tomography angiography without noninvasive fractional flow reserve
- Invasive coronary angiography
- Other noninvasive functional tests
| Relevant outcomes include:
- Test accuracy
- Test validity
- Morbid events
- Quality of life
- Resource utilization
- Treatment-related morbidity
|
BACKGROUND
Stable Ischemic Heart Disease
Coronary artery disease (CAD) is a significant cause of morbidity and mortality. Various epidemiologic risk factors have been well studied. Evaluation of obstructive CAD involves quantifying arterial stenoses to determine whether significant narrowing is present. Lesions with stenosis more than 50% to 70% in diameter accompanied by symptoms are generally considered significant. It has been suggested that coronary computed tomography angiography (CCTA) or other noninvasive functional cardiac testing may help rule out CAD and avoid invasive coronary angiography (ICA) in patients with a low clinical likelihood of significant CAD. However, ICAs are frequently unnecessary in patients with suspected stable ischemic heart disease (SIHD), as evidenced by low diagnostic yields for significant obstructive CAD. Patel et al (2010) found that from a sample of over 132 000 ICAs, 48.8% of elective ICAs performed in patients with stable angina did not detect obstructive CAD (left main stenosis ≥50% or ≥70% in a major epicardial or branch >2.0 mm in diameter).1, ICA is clinically useful when patients with stable angina have failed optimal medical therapy and may benefit from revascularization. A noninvasive imaging test performed before ICA as a gatekeeper, which can distinguish candidates who may benefit from early revascularization (eg, patients with unprotected left main stenosis ≥50% or hemodynamically significant disease) from those unlikely to benefit, could avoid unnecessary invasive procedures and their potential adverse consequences. Moreover, for the large majority of patients with SIHD, revascularization offers no survival advantage over medical therapy; few might benefit from ICA if they have not first failed optimal medical therapy.2,
Clinical Risk Prediction for Stable Ischemic Heart Disease
The 2012 collaborative medical association guidelines for the diagnosis and management of patients with stable heart disease list several class I recommendations on the use of noninvasive testing in patients with suspected SIHD.3, A class I recommendation indicates that a test should be performed. In general, patients with at least intermediate risk (10%-90% risk by standard risk prediction instruments) are recommended to have some type of test, the choice depending on the interpretability of the electrocardiogram, the capacity to exercise, and presence of comorbidity.
Clinical prediction scores or models have been developed to help estimate the pretest probability of CAD in individuals with stable chest pain. Diamond and Forrester (1979) developed the original version of a commonly cited clinical prediction model based on age, sex, and type of pain symptoms.,4,Genders et al (2011) further studied and extended the model.5,Wasfy et al (2012)compared it to the Duke Clinical Score.6, Versteylen et al (2011) published a comparison of clinical prediction results for the Diamond and Forrester (1979) model, the Framingham risk score, the PROCAM risk score, and the SCORE risk estimation model.7,Min et al (2015) published another model,8, and in 2016 a COD consortium developed an online calculator.9,10,
Gatekeepers to Invasive Coronary Angiography
Imposing an effective noninvasive gatekeeper strategy with one or more tests before planned ICA to avoid unnecessary procedures is compelling. The most important characteristic of a gatekeeper test is its ability to accurately identify and exclude clinically insignificant disease where revascularization would offer no potential benefit. From a diagnostic perspective, an optimal strategy would result in few false-negative tests while avoiding an excessive false-positive rate—it would provide a low post-test probability of significant disease. Such a test would then have a small and precise negative likelihood ratio and high negative predictive value. An effective gatekeeper would decrease the rate of ICA while increasing the diagnostic yield (defined by the presence of obstructive CAD on ICA). At the same time, there should be no increase in major adverse cardiac events. A clinically useful strategy would satisfy these diagnostic performance characteristics and impact the outcomes of interest. Various tests have been proposed as potentially appropriate for a gatekeeper function before planned ICA, including CCTA, magnetic resonance imaging, single-photon emission computed tomography, positron emission tomography, and stress echocardiography. More recently, adding noninvasive measurement of fractional flow reserve (FFR) using CCTA has been suggested, combining functional and anatomic information.
Fractional Flow Reserve
Invasively measured FFR evaluates the severity of ischemia caused by coronary artery obstructions and can predict when revascularization may be beneficial.11,12,13, FFR has not been used as a diagnostic test for ischemic heart disease, but as a test to evaluate the degree of ischemia caused by stenosis.
Invasive FFR is rarely used in the U. S. to guide percutaneous coronary intervention (PCI). Pothineni et al (2016), using the National Inpatient Sample, reported that 201 705 PCIs were performed in 2012, but just 21 365 FFR procedures.14, Assuming the intention of FFR is to guide PCI, it would represent just 4.3% of PCI procedures. Whether noninvasively obtained FFR will influence decisions concerning ICA, over and above anatomic considerations, is therefore important to establish empirically.
Randomized controlled trials and observational studies have demonstrated that FFR-guided revascularization can improve cardiovascular outcomes, reduce revascularizations, and decrease costs.15, For example, the Fractional Flow Reserve versus Angiography for Multivessel Evaluation trial randomized 1005 patients with multivessel disease and planned PCI.13,16, At 1 year, compared with PCI guided by angiography alone, FFR-guided PCI reduced the number of stents placed by approximately 30%, followed by lower rates (13.2% vs. 18.3%) of major cardiovascular adverse events (myocardial infarction, death, repeat revascularization) and at a lower cost. The clinical benefit persisted through 2 years, although by 5 years, event rates were similar between groups.17,
European guidelines (2013) for stable CAD have recommended that FFR be used "to identify hemodynamically relevant coronary lesion(s) when evidence of ischaemia is not available" (class Ia), and "[r]evascularization of stenoses with FFR <0.80 is recommended for patients with angina symptoms or a positive stress test."18, Other guidelines (2014) have recommended using "FFR to identify haemodynamically relevant coronary lesion(s) in stable patients when evidence of ischaemia is not available" (class Ia recommendation).19,The U.S. guidelines (2012) have stated that an FFR of 0.80 or less provides level Ia evidence for revascularization for "significant stenoses amenable to revascularization and unacceptable angina despite guideline directed medical therapy."3,Also, the importance of FFR in decision making appears prominently in the 2017 appropriate use criteria for coronary revascularization in patients with SIHD.20,
Measuring FFR during ICA can be accomplished by passing a pressure-sensing guidewire across a stenosis. Coronary hyperemia (increased blood flow) is then induced and pressure distal and proximal to the stenosis is used to calculate flow across it. FFR is the ratio of flow in the presence of a stenosis to flow in its absence. FFR levels less than 0.75 to 0.80 are considered to represent significant ischemia while those 0.94 to 1.0 are considered normal. Measurement is valid in the presence of serial stenoses, is unaffected by collateral blood flow,21,and reproducibility is high.22, Potential complications include adverse events related to catheter use such as vessel wall damage (dissection); the time required to obtain FFR during a typical ICA is less than 10 minutes.
FFR using CCTA requires at least 64-slice CCTA and cannot be calculated when images lack sufficient quality23, (11% to 13% in recent studies24,25,26,27,), eg, in obese individuals (body mass index, >35 kg/m2). The presence of dense arterial calcification or an intracoronary stent can produce significant beam-hardening artifacts and may preclude satisfactory imaging. The presence of an uncontrolled rapid heart rate or arrhythmia hinders the ability to obtain diagnostically satisfactory images. Evaluation of the distal coronary arteries is generally more difficult than the visualization of the proximal and mid-segment coronary arteries due to greater cardiac motion and the smaller caliber of coronary vessels in distal locations.
Noninvasive Fractional Flow Reserve Measurement
FFR can be modeled noninvasively using images obtained during CCTA28,CCTA (HeartFlow software termed FFRCT; Siemens cFFR). The process involves constructing a digital model of coronary anatomy and calculating FFR across the entire vascular tree using computational fluid dynamics. FFR using CCTA can also be used for "virtual stenting" to simulate how stent placement would be predicted to improve vessel flow.29,
Only HeartFlow FFRCT software has been cleared by the U.S. Food and Drug Administration (FDA). Imaging analyses require transmitting data to a central location for analysis, taking 1 to 3 days to complete. Other prototype software is workstation-based with onsite analyses.
Regulatory Status
In November 2014, FFRCT simulation software (HeartFlow) was cleared for marketing by the FDA through the de novo 510(k) process (class II, special controls; FDA product code: PJA). In January 2016, the FFRCT v2.0 device was cleared through a subsequent 510(k) process.
HeartFlow FFRCT post-processing software is cleared:
"for the clinical quantitative and qualitative analysis of previously acquired Computed Tomography [CT] DICOM [Digital Imaging and Communications in Medicine] data for clinically stable symptomatic patients with coronary artery disease. It provides FFRCT [fractional flow reserve using coronary computed tomography angiography], a mathematically derived quantity, computed from simulated pressure, velocity and blood flow information obtained from a 3D computer model generated from static coronary CT images. FFRCT analysis is intended to support the functional evaluation of coronary artery disease. The results of this analysis [FFRCT] are provided to support qualified clinicians to aid in the evaluation and assessment of coronary arteries. The results of HeartFlow FFRCT are intended to be used by qualified clinicians in conjunction with the patient's clinical history, symptoms, and other diagnostic tests, as well as the clinician's professional judgment."
Related Policies
- Adult Cardiac Imaging Policy (Policy #149 in the Radiology Section)
Policy:
(NOTE: For Medicare Advantage, please refer to the Medicare Coverage Section below for coverage guidance.)
1. The use of noninvasive fractional flow reserve following a positive coronary computed tomography angiography is considered medically necessary to guide decisions about the use of invasive coronary angiography in members with stable chest pain at intermediate risk of coronary artery disease (i.e., suspected or presumed stable ischemic heart disease).
2. The use of noninvasive fractional flow reserve not meeting the criteria outlined above is considered investigational.
Policy Guidelines: (Information to guide medical necessity determination based on the criteria contained within the policy statements above.)
Fractional flow reserve using coronary computed tomography angiography requires at least 64-slice coronary computed tomography angiography and cannot be calculated when images lack sufficient quality (HeartFlow, 2013) (11% to 13% in recent studies; Koo et al, 2011; Min et al, 2012; Nakazato et al, 2013; Nørgaard et al, 2014), eg, in obese individuals (eg, body mass index, >35 kg/m2). The presence of dense arterial calcification or an intracoronary stent can produce significant beam-hardening artifacts and may preclude satisfactory imaging. The presence of an uncontrolled rapid heart rate or arrhythmia hinders the ability to obtain diagnostically satisfactory images. Evaluation of the distal coronary arteries is generally more difficult than visualization of the proximal and mid-segment coronary arteries due to greater cardiac motion and the smaller caliber of coronary vessels in distal locations.
Medicare Coverage:
There is no National Coverage Determination (NCD) or Local Coverage Determination (LCD) for jurisdiction JL for this service. Therefore, Medicare Advantage will follow the Horizon BCBSNJ Medical Policy.
[RATIONALE: This policy was created in 2017 and has been updated regularly with searches of the PubMed database. The most recent literature update was performed through March 9, 2020.
Evidence reviews assess whether a medical test is clinically useful. A useful test provides information to make a clinical management decision that improves the net health outcome. That is, the balance of benefits and harms is better when the test is used to manage the condition than when another test or no test is used to manage the condition.
The first step in assessing a medical test is to formulate the clinical context and purpose of the test. The test must be technically reliable, clinically valid, and clinically useful for that purpose. Evidence reviews assess the evidence on whether a test is clinically valid and clinically useful. Technical reliability is outside the scope of these reviews, and credible information on technical reliability is available from other sources.
Coronary Computed Tomography Angiography with Selective Noninvasive Fractional Flow Reserve
Clinical Context and Test Purpose
The purpose of selective noninvasive fractional flow reserve (FFR) using coronary computed tomography angiography (CCTA; collectively FFR-CT) in patients with stable chest pain who have suspected stable ischemic heart disease (SIHD) and who are being considered for invasive coronary angiography (ICA) is to select patients who may be managed safely with observation only, instead of undergoing ICA in the short term.
The question addressed in this policy is: Does noninvasive FFR-CT guide decisions to use or not use ICA and improve net health outcomes in patients with stable chest pain or suspected SIHD?
The following PICO was used to select literature to inform this policy.
Patients
The population of interest is patients with stable chest pain at intermediate risk of coronary artery disease (CAD; ie, with suspected or presumed SIHD) who are being considered for ICA. Patients may have undergone prior noninvasive testing and been treated for presumed stable angina.
Interventions
The test being considered is CCTA with selective FFR-CT when CCTA shows evidence of coronary artery stenosis. The setting is a general cardiology practice for patients undergoing nonemergent chest pain evaluation.
Comparators
The following tests are currently being used. Patients may receive CCTA, which may be performed alone without FFR. They may proceed directly to ICA. Conventional noninvasive imaging tests providing functional information, including myocardial perfusion imaging (MPI) using single-photon emission computed tomography (SPECT), stress echocardiography (SECHO), and cardiac positron emission tomography (PET), may be used before ICA. Cardiovascular magnetic resonance imaging (MRI) is also an option. The setting is a general cardiology practice for patients undergoing nonemergent chest pain evaluation.
Outcomes
The final outcomes of interest include ICA rates, ICA without obstructive CAD, major adverse cardiovascular events (MACE), and adverse events attributed to testing and treatment. Rates of ICA and treatment-related morbidity are typically short-term (eg, ≤3 months). Also, rates of subsequent ICA, treatment-related morbidity, MACE, quality of life, and resource utilization ascertained over a period of 1 to 3 years are also of interest.
The intermediate outcome of interest is the ability of the test to distinguish clinically significant CAD for which revascularization may provide benefit.
Study Selection Criteria
For the evaluation of clinical validity of this test, studies that meet the following eligibility criteria were considered:
- Reported on the accuracy of the marketed version of the technology (including any algorithms used to calculate scores)
- Included a suitable reference standard (describe the reference standard)
- Patient/sample clinical characteristics were described
- Patient/sample selection criteria were described
Technically Reliable
Assessment of technical reliability focuses on specific tests and operators and requires review of unpublished and often proprietary information. Review of specific tests, operators, and unpublished data are outside the scope of this policy, and alternative sources exist. This policy focuses on the clinical validity and clinical utility.
Clinically Valid
A test must detect the presence or absence of a condition, the risk of developing a condition in the future, or treatment response (beneficial or adverse).
Studies Included in Fractional Flow Reserve-CT Systematic Reviews: Per-Patient Diagnostic Accuracy
Review of Evidence
Takx et al (2015) reported on a meta-analysis for which 26 studies contributed patient-level results that examined 5 non–FFR-CT imaging modalities (see Table 1).30,Wu et al (2016) and Danad et al (2017) each had 5 studies contributing results to 2 meta-analysesWu et al (2016)31,32,that evaluated the diagnostic accuracy of FFR-CT using patients as the unit of analysis. Only the U.S. Food and Drug Administration (FDA)-cleared HeartFlow software has been evaluated prospectively across multiple sites. Three small retrospective studies have reported per-patient performance characteristics for the prototype Siemens workstation-based software.33,34,35, The 3 HeartFlow FFRCT studies used successive software versions with reported improvement in specificity (from 54% to 79%) between versions 1.2 and 1.4.24,27,36, The Analysis of Coronary Blood Flow Using CT Angiography: Next Steps (NXT) Trial, the basis for device clearance by the FDA, was conducted at 11 sites in 8 countries (Canada, EU, Asia).27, Although not examined in the 2 included meta-analyses, subgroup analyses suggested little variation in results by sex and age.37, Effectively, the entirety of the data was obtained in patients of white or Asian descent; almost all patients were appropriate for testing according to the FDA clearance.
Danad et al (2017)
Danad et al (2017) included 23 studies published between January 2002 and February 2015 evaluating the diagnostic performance of CCTA, FFR-CT, SPECT, SECHO, MRI, or ICA compared with an invasive FFR reference standard.32, The 3 included FFR-CT studies used the HeartFlow software and had performed FFR in at least 75% of patients. A cutoff of 0.75 defined significant stenosis in 8 (32%) studies and the remainder 0.80 (the current standard used in all FFR-CT studies). Per-patient and per-vessel meta-analyses were performed. Study quality was assessed using QUADAS-238,; no significant biases were identified in FFR-CT studies, but a high risk of biased patient selection was judged in 10 (43.4%) of other studies. HeartFlow funded the Open Access publication; one author was a consultant to, and another was a cofounder.
On the patient level, MRI had the highest combined sensitivity (90%) and specificity (94%) for invasive FFR, but were estimated from only 2 studies (70 patients). FFR-CT had similar sensitivity, but lower specificity, and, accordingly, a lower positive likelihood ratio than MRI. The negative likelihood ratios were low (lower is better) for both FFR-CT and MRI; however, the confidence interval is narrower for FFR-CT due to larger sample for FFR-CT. CCTA had a slightly higher negative likelihood ratio. Results for the per-vessel area under the summary receiver operating characteristic curve were similar except for CCTA, for which per-patient results were considerably worse (eg, C statistic of 0.57 vs. 0.85). Reviewers noted heterogeneity in many estimates (eg, CCTA sensitivity, I2=80%). Finally, pooled results for some imaging tests included few studies (see Table 1 for detailed results).
Wu et al (2016)
Wu et al (2016) identified 7 studies (833 patients, 1377 vessels) comparing FFR-CT with invasively measured FFR from searches of PubMed, Cochrane, EMBASE, Medion, and meeting abstracts through January 2016.31, Studies included patients with established or suspected SIHD. In addition to the 3 FFR-CT studies pooled by Danad et al (2017), an additional study using HeartFlow technique (44 patients; 48 vessels) and 3 additional studies (180 patients; 279 vessels) using Siemens cFFR software (not FDA approved or cleared) were identified. An invasive FFR cutoff of 0.80 was the reference standard in all studies. Per-patient results reported in 5 studies were pooled and are reported in Table 1. All studies were rated at low risk of bias and without applicability concerns using the QUADAS-2 tool.38, Appropriate bivariate meta-analyses (accounting for correlated sensitivity and specificity) were used.
As expected given study overlap, FFR-CT performance characteristics were similar to those reported by Danad et al (2017), but with slightly higher specificity (see Table 1). The pooled per-vessel C statistic was lower (0.86) than the per-patient result (0.90). No evidence of publication bias was detected, but the number of studies was too small to assess adequately. Reviewers noted that in 2 studies FFR-CT results were uninterpretable in 12.0%27, and 8.2%39, of participants.
Takx et al (2015)
Takx et al (2015) identified studies reporting on the ability of perfusion computed tomography (CT), MRI, SECHO, PET, and SPECT to detect hemodynamically significant CAD as measured by ICA with invasive FFR.30, Studies published through May 2014 were eligible for inclusion; PubMed, EMBASE, and Web of Science were searched. QUADAS-2 was used to assess study quality38,; studies generally rated poorly on blinding of the index test result from the assessor and study population selection. Reviewers designated the negative likelihood ratio as the diagnostic characteristic of interest (ie, ability to exclude disease) noting that MPI (eg, MRI, SPECT, PET, or CT) has been proposed to be a gatekeeper to ICA. No funding was obtained for the review, and the study was registered on the International prospective register of systematic reviews40, (the 2 other meta-analyses were not).
The pooled negative likelihood ratios for MRI, PET, and perfusion CT were similar in magnitude (0.12 to 0.14; see Table 1) although the CI for PET was wide. Heterogeneity among studies included in the pooled patient-level results was considered high for PET (I2=84%), moderate for CT (I2=70%) and SPECT (I2=55%), and low for MRI (I2=0%) and SECHO (I2=0%). Publication bias, when able to be assessed, was not suspected. Concerning the ability to detect hemodynamically significant ischemia, reviewers concluded that "MPI with MRI, CT, or PET has the potential to serve as a gatekeeper for invasive assessment of hemodynamic significance by ICA and FFR." Studies of FFR-CT were not included in the analysis.
Table 1. Pooled Per-Patient Pooled Diagnostic Performance of Noninvasive Tests for Invasive FFR
| Test | Studies | N | Sensitivity
(95% CI), % | Specificity
(95% CI), % | C | LR+ (95% CI) | LR- (95% CI) |
| Danad et al (2017)32, |
| MRI | 2 | 70 | 90 (75 to 97) | 94 (79 to 99) | 0.94 | 10.3 (3.14 to 33.9) | 0.12 (0.05 to 0.30) |
| FFR-CT | 3 | 609 | 90 (85 to 93) | 71 (65 to 75) | 0.94 | 3.3 (1.78 to 6.25) | 0.16 (0.11 to 0.23) |
| CCTA | 4 | 694 | 90 (86 to 93) | 39 (34 to 44) | 0.57 | 1.5 (1.25 to 1.90) | 0.22 (0.10 to 0.50) |
| SECHO | 2 | 115 | 77 (61 to 88) | 75 (63 to 85) | 0.82 | 3.0 (1.94 to 4.65) | 0.34 (0.17 to 0.66) |
| SPECT | 3 | 110 | 70 (59 to 80) | 78 (68 to 87) | 0.79 | 3.4 (1.04 to 11.1) | 0.40 (0.19 to 0.83) |
| ICA | 2 | 954 | 69 (65 to 75) | 67 (63 to 71) | 0.75 | 2.5 (1.25 to 5.13) | 0.46 (0.39 to 0.55) |
| Wu et al (2016)31, |
| FFR-CT | 5 | 833 | 89 (85 to 93) | 76 (64 to 84) | 0.90 | 3.7 (2.41 to 5.61) | 0.14 (0.09 to 0.21) |
| Takx et al (2015)30, |
| MRI | 10 | 798 | 89 (86 to 92) | 87 (83 to 90) | 0.94 | 6.3 (4.88 to 8.12) | 0.14 (0.10 to 0.18) |
| PCT | 5 | 316 | 88 (82 to 92) | 80 (73 to 86) | 0.93 | 3.8 (1.94 to 7.40) | 0.12 (0.04 to 0.33) |
| SECHO | 4 | 177 | 69 (56 to 79) | 84 (75 to 90) | 0.83 | 3.7 (1.89 to 7.15) | 0.42 (0.30 to 0.59) |
| SPECT | 8 | 533 | 74 (67 to 79) | 79 (74 to 83) | 0.82 | 3.1 (2.09 to 4.70) | 0.39 (0.27 to 0.55) |
| PET | 2 | 224 | 84 (75 to 91) | 87 (80 to 92) | 0.93 | 6.5 (2.83 to 15.1) | 0.14 (0.02 to 0.87) |
CCTA: coronary computed tomography angiography; CI: confidence interval; FFR-CT: fractional flow reserve using coronary computed tomography angiography; ICA: invasive coronary angiography; LR: likelihood ratio; MRI: magnetic resonance imaging; PCT: perfusion computed tomography; PET: positron emission tomography; SECHO: stress echocardiography; SPECT: single-photon emission computed tomography.
Section Summary: Clinically Valid
Three studies including 609 patients have evaluated the diagnostic accuracy of the FDA-cleared HeartFlow software. Software used in successive studies was also revised to improve performance characteristics, particularly specificity. For example, using an earlier software version, the noninvasive fractional flow reserve derived from computed tomography angiography for coronary lesions of intermediate stenosis severity trial reported a specificity of 54%.41, Accordingly, pooled results from the Danad et al (2017) systematic review must be interpreted carefully. Also, there is some uncertainty in the generalizability of results obtained in these studies conducted under likely controlled conditions (eg, data from the NXT Trial27, forming the basis for the FDA clearance).
Given the purpose to avoid ICA, the negative likelihood ratio, or how a negative result might dissuade a clinician from proceeding to ICA, is of primary interest, ie, excluding a patient with vessels having a high FFR from ICA. While CIs are relatively wide and overlapping, the negative likelihood ratio estimates of FFR-CT for excluding physiologically significant coronary stenoses tended to be lower (ie, better) than CCTA alone, SECHO, SPECT, and ICA. Only MRI yielded a similarly low or lower negative likelihood ratio than FFR-CT.
Clinically Useful
A test is clinically useful if the use of the results informs management decisions that improve the net health outcome of care. The net health outcome can be improved if patients receive correct therapy, or more effective therapy, or avoid unnecessary therapy, or avoid unnecessary testing.
Direct Evidence
Direct evidence of clinical utility is provided by studies that have compared health outcomes for patients managed with and without the test. Because these are intervention studies, the preferred evidence would be from randomized controlled trials (RCTs).
Review of Evidence
Two prospective comparative studies were identified:ne 1 prospective nonrandomized study that compared an FFR-CT strategy (CCTA plus noninvasive FFR measurement when requested or indicated) with ICA and 1 RCT that examined CCTA as a gatekeeper to ICA (see Tables 2 and 3). Also identified were 1 prospective cohort study, along with 2 retrospective cohort studies and 1 case series of patients referred for CCTA, which included FFR-CT evaluation.
PLATFORM Study
The Prospective LongitudinAl Trial of FFRCT: Outcome and Resource Impacts (PLATFORM) study compared diagnostic strategies with or without FFR-CT in patients with suspected stable angina but without known CAD.42,43, The study was conducted at 11 EU sites. All testing was nonemergent. Patients were divided into 2 strata, according to whether the test planned before study enrollment was: (1) noninvasive or (2) ICA (the patient population of interest in this policy). Patients were enrolled in consecutive cohorts (see Table 2), with the first cohort undergoing a usual care strategy followed by a second cohort provided CCTA with FFR-CT performed when requested (recommended if stenoses ≥30% were identified). Follow-up was scheduled for 90 days and 6 and 12 months after entry (99.5% of patients had 1-year follow-up data). Funding was provided by HeartFlow, and multiple authors reported receiving fees, grants, and/or support from HeartFlow. Data analyses were performed by the Duke Clinical Research Institute.
ICA without obstructive disease ("no stenosis ≥ 50% by core laboratory quantitative analysis or invasive FFR < 0.80") at 90 days was the primary endpoint in patients with planned invasive testing."no stenosis ≥ 50% by core laboratory quantitative analysis or invasive FFR < 0.80." Secondary endpoints included ICA without obstructive disease following planned noninvasive testing, and (1) MACE at 1 year defined as a composite of all-cause mortality, myocardial infarction (MI), and urgent revascularization, and (2) MACE and vascular events within 14 days. quality of life was evaluated using the Seattle Angina Questionnaire, and EuroQol-5D (5-item and 100-point visual analog scale). CCTA studies were interpreted by site investigators; quantitative coronary angiography measurements were performed at a central laboratory, as was FFR-CT. Cumulative radiotherapy was also assessed. A sample size of 380 patients in the invasive strata yielded a 90% power to detect a 50% decrease in the primary endpoint given a 30% event rate (ICA without obstructive disease) with a usual care strategy and a dropout rate up to 10%.
ICA was planned in 380 participants, of whom 193 (50.8%) had undergone prior noninvasive testing. The mean pretest probability in the planned ICA strata was approximately 50% (51.7% and 49.4% in the 2 groups). FFR-CT was requested in 134 patients and successfully obtained in 117 (87.3%) of 134 in the FFR-CT group. At 90 days, 73.3% of those in the usual care group had no obstructive findings on ICA compared with 12.4% in the FFR-CT group based on core laboratory readings (56.7% and 9.3% based on site readings) (see Table 3). The difference was similar in a propensity-matched analysis of a subset of participants (n=148 from each group, or 78% of the entire sample). Prior noninvasive testing did not appear associated with nonobstructive findings. MACE rates were low and did not differ between strategies. Mean level of radiation exposure through 1 year was also similar in the usual care group (10.4 mSv) and the planned ICA group (10.7 mSv). No differences in quality of life were found between groups.44,
Results of the PLATFORM study supported the notion that, in patients with planned ICA, FFR-CT can decrease the rate of ICAs and unnecessary procedures (finding no significant obstructive disease) and that FFR-CT may provide clinically useful information to physicians and patients (see Table 3). Study limitations include a nonrandomized design; high rate of no obstructive disease with a usual care strategy (73.3%), which was higher than the 30% rate assumed in the sample size estimates; and a sample size that was small with respect to evaluating adverse cardiac events. Although finding a large effect in patients with planned invasive testing, the nonrandomized design limits causal inferences and certainty in the magnitude of the effect. The propensity-matched analysis (in a matched subset) offers some reassurance, but the sample size was likely too small to provide robust results.
Coronary Artery Disease Management (CAD-Man) Trial
Dewey et al (2016) conducted the Coronary Artery Disease Management (CAD-Man) trial, a single-center, parallel-group assignment trial examining CCTA as a gatekeeper to ICA in patients with atypical angina or chest pain and suspected CAD who were indicated for ICA.45, Patients were randomized to direct ICA or to ICA, only if a prior CCTA was positive (a stenosis ≥70% stenosis in any vessel or ≥50% in the left main coronary artery) (see Table 2). The trialists reported that when obstructive disease was suspect following CCTA, late enhancement MRI was performed to evaluate the extent of viable myocardium (completed in 17 patients) to guide revascularization; however, the study protocol clarified that MRI was not used for decisions to proceed to ICA. A major procedural complication (death, stroke, MI, or event requiring >24-hour hospitalization) within 24 hours was the primary outcome; secondary outcomes included ICA with obstructive CAD (diagnostic yield), revascularizations, and MACE during long-term follow-up. The trial was performed in Germany. Patients were excluded if they had evidence of ischemia or signs of MI, and just over half (56.5%) were inpatients at the time of enrollment. Obstructive disease was defined as "at least one 50% diameter stenosis in the left main coronary artery or at least one 70% diameter stenosis in other coronary arteries." Allocation concealment appeared adequate, but the trial was unblinded owing to the nature of the intervention. Also, the mean pretest probability of CAD at baseline was higher in the ICA-only arm (37.3% vs. 31.3%; see Table 2). The research was supported by public funding.
ICAs were reduced by 85.6% in the CCTA arm and by 80.9% for ICA with no obstructive disease. A major procedural complication (the primary outcome) occurred in a single patient undergoing CCTA. Percutaneous coronary interventions (PCIs) were less frequent when CCTA was performed (9.6% vs. 14.2%; p<.001) (see Table 3). Over a median follow-up of 3.3 years, MACE rates were similar in the trial arms (4.2% in the CCTA group vs. 3.7% with ICA; adjusted hazard ratio, 0.90; 95% CI, 0.30 to 2.69). In the CCTA arm, there was 1 death, 2 patients with unstable angina, and 6 revascularizations; in the ICA arm, there was 1 MI, 1 stroke, and 5 revascularizations.
The trial demonstrated that CCTA as a gatekeeper to planned ICA could avoid a large number of procedures, a corresponding increase in the diagnostic yield, and fewer revascularizations (see Table 3). Of note, the prevalence of obstructive CAD found on ICA in this study population was 13% (43/334 eligible for primary outcome analysis), which is lower than the prevalence of obstructive CAD in the PLATFORM population (26.7%). Thus, the subset of individuals who went on to ICA following CCTA findings of obstructive CAD was 20 (12%) of 167 eligible for primary outcome analysis, and only 3 (1.7%) were found to have no obstructive CAD on ICA. MACE rates did not differ between arms. The trial was powered neither to detect a difference nor to assess noninferiority-implications of the absence of a difference are limited. Finally, although the patient population included those scheduled for elective ICA, it was heterogeneous, including those with recent onset and longer-standing chest pain. The single-center nature of the trial is an additional limitation; a subsequent Diagnostic Imaging Strategies for Patients with Stable Chest Pain and Intermediate Risk of Coronary Disease trial is ongoing.
Table 2. Characteristics of Comparative Studies
| Characteristics | Nonrandomized | Randomized |
 | PLATFORM | CAD-Man |
 | ICA | FFR-CT | ICA | CCTA |
 | (n=187) | (n=193) | (n=162) | (n=167) |
| Age (SD), y | 63.4 (10.9) | 60.7 (10.2) | 60.4 (11.4) | 60.4 (11.3) |
| Female, n (%) | 79 (42.2) | 74 (38.3) | 88 (52.7) | 78 (48.1) |
| Race/ethnic minority, n (%) | 2 (1.1) | 1 (0.5) |  |  |
| Pretest probability obstructive CAD (SD), % | 51.7 (16.7) | 49.4 (17.2) | 37.3 (24.8) | 31.3 (21.1) |
| Angina, n (%) |  |  |  |  |
| Typical | 52 (27.8) | 45 (23.3) |  |  |
| Atypical | 122 (65.2) | 142 (73.6) | 79 (48.8) | 65 (38.9) |
| Noncardiac | 12 (7.0) | 5 (2.6) | 80 (49.4) | 97 (58.1) |
| Other chest discomfort |  |  | 3 (1.8) | 5 (3.0) |
| Prior noninvasive testing, n (%) | 92 (49.2) | 101 (52.3) | 84 (50.3) | 92 (56.8) |
| Diabetes, n (%) | 36 (19.3) | 30 (15.5) | 30 (18.5) | 15 (9.0) |
| Current smoker |  |  | 34 (21.0) | 41 (24.5) |
| Current or past smoker | 103 (55.1) | 101 (52.3) | 85 (52.4) | 88 (52.6) |
Adapted from Douglas et al (2015, 2016)42,NA43, and Dewey et al (2016).45,
CAD: coronary artery disease; CCTA: coronary computed tomography angiography; FFR-CT: fractional flow reserve using coronary computed tomography angiography; ICA: invasive coronary angiography; SD: standard deviation.
Table 3. Results of Comparative Studies
| Outcomes | Nonrandomized | Randomized |
 | PLATFORM | CAD-Man |
 | ICA | FFR-CT | ICA | CCTA |
 | (n=187) | (n=193) | (n=162) | (n=167) |
| Noninvasive FFR-CT, n (%) |  |  |  |  |
| Requested |  | 134 (69.4) |  |  |
| Successfully performed |  | 117 (60.1) |  |  |
| ICA no obstructive disease, n (%) | 137 (73.3) | 24 (12.4) | 137 (84.5) | 6 (3.6) |
| Absolute difference (95% CI), % | 60.8 (53.0 to 68.7) | 80.9 (74.6 to 87.2) |
| ICA, n (%) | 187 (100) | 76 (39.4) | 162 (100) | 24 (14.4) |
| Absolute difference (95% CI), % | 60.6 (53.7 to 67.5) | 85.6 (80.3 to 90.9) |
| Revascularization, n (%) |  |  |  |  |
| PCI | 49 (26.2) | 55 (28.5) |  |  |
| CABG | 18 (9.6) | 10 (5.2) |  |  |
| Any | 67 (35.8) | 65 (33.7) | 23 (14.2) | 16 (9.6) |
| 1-year outcomes, n (%) |  |  |  |  |
| MACEa | 2 (1.1) | 2 (1.0) |  |  |
| MACEb |  |  | 6 (3.7) | 7 (4.2) |
Adapted from Douglas et al (2015, 2016)42,NA43, and Dewey et al (2016).45,
CABG: coronary artery bypass grafting; CCTA: coronary computed tomography angiography; CI: confidence interval; FFR-CT: fractional flow reserve using coronary computed tomography angiography; ICA: invasive coronary angiography; MACE: major adverse cardiovascular events; PCI: percutaneous coronary intervention.
a Death, myocardial infarction, unplanned urgent revascularization.
b Cardiac death, myocardial infarction, stroke, unstable angina, any revascularization.
Jensen et al (2018) (Prospective Cohort)
Jensen et al (2018) reported on a single-institution study of 774 consecutive individuals with suspicion of CAD referred for nonemergent ICA or CCTA.46, Subjects were analyzed in 2 groups: a low-to-intermediate–risk group accounting for 76% of patients with a 31% mean pretest probability of CAD and a high-risk group accounting for 24% of patients with a 67% mean pretest probability of CAD. Among the 745 who received CCTA, FFR-CT was selectively ordered in 28% of patients overall (23% in the low-to-intermediate–risk group, 41% in the high-risk group). CCTA was considered inconclusive in 3% of subjects, and, among those with conclusive CCTA, FFR-CT yielded few inconclusive results, with less than 3% of cases. During a minimum 90-day follow-up, the combined testing strategy of selective FFR-CT following CCTA resulted in avoiding ICA in 91% of low-to-intermediate–risk and 75% of high-risk individuals. None of the patients who avoided ICA based on CCTA with selective FFR-CT were associated with serious clinical adverse events over an average of 157 days of follow-up.
Nørgaard et al (2017) (Retrospective Cohort)
Nørgaard et al (2017) reported on results from symptomatic patients referred for CCTA at a single-center in Denmark from May 2014 to April 2015.47, All data were obtained from medical records and registries; the study was described as a "review" of diagnostic evaluations and was apparently retrospectively conducted. Follow-up through 6 to 18 months was ascertained. From 1248 referred patients, 1173 underwent CCTA; 858 received medical therapy, 82 underwent ICA, 44 MPI, and 189 FFR-CT (185 [98%] obtained successfully). Of the 185 individuals who successfully obtained FFR-CT, FFR-CT demonstrated values of 0.80 or less in 1 or more vessels in 57 (31%) patients, and 49 (86%) went on to ICA; whereas of the 128 with higher FFR-CT values, only 5 (4%) went on to ICA. Assuming ICA was planned for all patients undergoing FFR-CT, these results are consistent with FFR-CT being able to decrease the rate of ICA. However, implications are limited by the retrospective design, performance at a single-center, and lack of a comparator arm including one for CCTA alone.
Lu et al (2017) Retrospective Cohort
Lu et al (2017) retrospectively examined a subgroup referred to ICA48, from the completed PROspective Multicenter Imaging Study for Evaluation of Chest Pain (PROMISE) trial. PROMISE was a pragmatic trial comparing CCTA with functional testing for the initial evaluation of patients with suspected SIHD.49, Of 550 participants referred to ICA within 90 days, 279 were not considered for the analyses due to CCTA performed without nitroglycerin (n=139), CCTA not meeting slice thickness guidelines (n=90), or nondiagnostic studies (n=50). Of the remaining 271 patients, 90 scans were inadequate to obtain FFR-CT, leaving 181 (33%) of those referred to ICA for analysis. Compared with those excluded, patients in the analytic sample were less often obese, hypertensive, diabetic, minority, or reported a CAD equivalent symptom. The 2 groups had similar pretest probabilities of disease, revascularization rates, and MACE, but the distribution of stenoses in the analytic sample tended to be milder (p=.06). FFR-CT studies were performed in a blinded manner and not available during the conduct of PROMISE for decision making.
Severe stenosis (≥70%) or left main disease (≥50%) were present in 110 (66%) patients by CCTA result and in 54% by ICA. Over a 29-month median follow-up, MACE (death, nonfatal MI, hospitalization for unstable angina) or revascularization occurred in 51% of patients (9% MACE, 49% revascularization). A majority (72%) of the sample had at least 1 vessel with an FFR-CT of 0.80 or less, which was also associated with a higher risk of revascularization but with a wide CI (hazard ratio=5.1; 95% CI, 2.6 to 11.5). If reserved for patients with an FFR-CT of 0.80 or less, ICAs might have been avoided in 50 patients (ie, reduced by 28%) and the rate of ICA without 50% or more stenosis from 27% (calculated 95% CI, 21% to 34%) to 15% (calculated 95% CI, 10% to 23%). If the 90 patients whose images were sent for FFR-CT but were unsatisfactory proceeded to ICA—as would have occurred in practice—the rate of ICA might have decreased by 18% and ICA without significant stenosis from 31% to 25%.
The authors suggested that when CCTA is used as the initial evaluation for patients with suspected SIHD, adding FFR-CT could have decreased the referral rate to ICA in PROMISE from 12.2% to 9.5%, or close to the 8.1% rate observed in the PROMISE functional testing arm. They also noted the similarity of their findings to PLATFORM and concluded, "In this hypothesis-generating study of patients with stable chest pain referred to ICA after [C]CTA, we found that adding FFRCT may improve the efficiency of referral to ICA, addressing a major concern of an anatomic [C]CTA strategy. FFRCT has incremental value over anatomic [C]CTA in predicting revascularization or major adverse cardiovascular events."
This retrospective observational subgroup analysis from PROMISE would suggest that when CCTA is the initial noninvasive test for the evaluation of suspected SIHD, FFR-CT before ICA has the potential to reduce unnecessary ICAs and increase the diagnostic yield. However, study limitations and potential generalizability are important to consider. First, analyses included only a third of CCTA patients referred to ICA, and some characteristics of the excluded group differed from the analytic sample. Second, conclusions assume that an FFR-CT greater than 0.80 will always dissuade a physician from recommending ICA and even in the presence of severe stenosis (eg, ≥70% in any vessel or ≥50% in the left main), or almost half (46%) of patients with an FFR-CT greater than 0.80. Finally, estimates including patients with either nondiagnostic CCTA studies (n=50) or studies inadequate for calculating FFR-CT (n=90) are more appropriate because in practice those patients would most likely proceed to ICA. Accordingly, the estimates are appropriately considered upper bounds for what might be seen in practice. It is also important to note that in strata of the PLATFORM trial enrolling patients for initial noninvasive testing (not planned ICA), ICA was more common following CCTA and contingent FFR-CT than following usual care (18.3% vs. 12.0%) and ICA, with no obstructive disease more frequent in the FFR-CT arm (12.5% vs. 6.0%).
The ADVANCE Registry Case Series
Patel et al (2020) conducted a registry study on the 1-year medical practice and clinical outcomes of FFRCT for patients in the international Assessing Diagnostic Value of Non-Invasive FFRCT in Coronary Care (ADVANCE) registry.50, Patients suspected of having CAD and with atherosclerosis identified by CCTA (N=5083 from 38 international sites) were prospectively enrolled in the registry from July 15, 2015 to October 20, 2017. Investigators recorded demographics, symptoms, CCTA and FFRCT findings, treatment plans, and clinical outcomes through 1 year, and these were then adjudicated by a blinded central laboratory. At 1 year, investigators had follow-up data from 4737 (93.2%) patients with FFRCT. Outcomes, detailed in Table 4, were revascularization, major adverse cardiac events, and time to first event (all-cause death or MI), and time to first event (cardiovascular death or MI). The 1-year outcomes showed low event rates in all patients; lower major adverse cardiac events and significantly lower cardiovascular death or MI were found in patients with an FFRCT >0.80 (negative) compared with those with positive (abnormal) FFRCT.
Table 4. 1-Year Outcomes From the ADVANCE Registry (N=4,634)
 | Revascularization | MACE | Time to first event |
 |  |  | All-cause death or MI | Cardiovascular death or MI |
FFRCT ≤0.80
(n=3145, 66.39%) | n=1208 (38.40%) | n=43 (1.37%) | n=38 (1.20%) | n=25 (0.80%) |
FFRCT >0.80
(n=1592, 33.61%) | n=89 (5.60%) | n=12 (0.75%) | n=10 (0.60%) | n=3 (0.20%) |
| RR | 6.87 | 1.81 | 1.92 | 4.22 |
| 95% CI | 5.59-8.45 | 0.96-3.43 | 0.96-3.85 | 1.28-13.95 |
| p-value | <.001 | .06 | .06 | .01 |
Source: Patel et al (2020)50,
CI: confidence interval; FFRCT: fractional flow reserve derived from coronary computed tomography angiography; MACE: major adverse cardiac events; MI: myocardial infarction; RR: relative risk.
Chain of Evidence
Indirect evidence on clinical utility rests on clinical validity. If the evidence is insufficient to demonstrate test performance, no inferences can be made about clinical utility.
Diagnostic performance can offer indirect evidence of clinical utility, assuming providers act according to a test result. As previously noted, an effective gatekeeper strategy must be able to decrease the probability of disease (rule out) sufficiently that a planned ICA would not be performed. Ruling out the disease is a function of the negative likelihood ratio that defines the degree to which a negative test decreases the posttest odds (and probability) of disease. The steps in the logic are illustrated in Figure 1.
Figure 1. Pathway for Clinical Use of FFR-CT to Support Clinical Utility
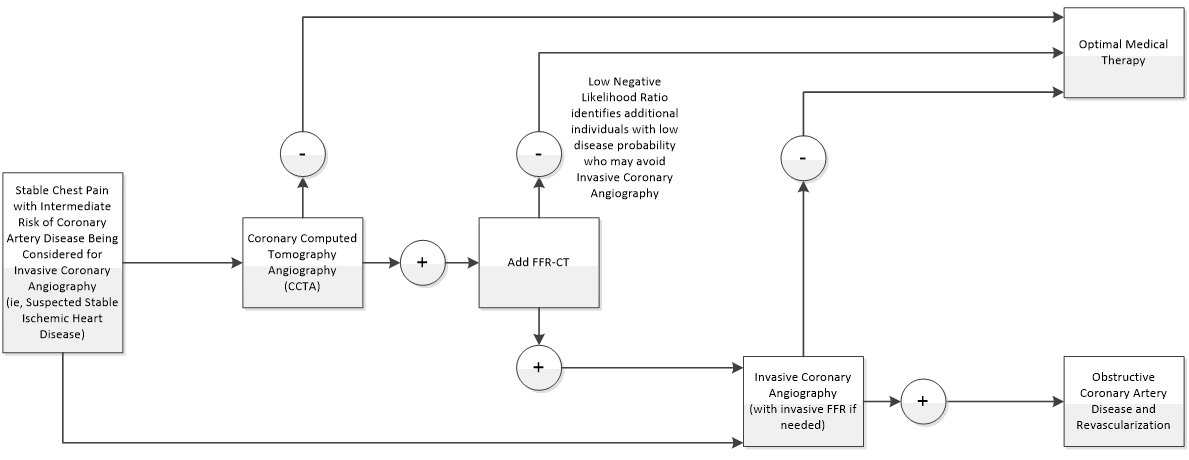
FFR-CT: fractional flow reserve using coronary computed tomography angiography.
Table 5 illustrates how a negative test would lower the probability of a hemodynamically significant obstruction from pretest probabilities of 0.25, 0.50, or 0.75 for the various tests examined in the meta-analyses. For example, according to the results of Danad et al (2017), if the pretest probability was 0.50, following a negative CCTA study the posttest probability would be 0.18. In contrast, beginning with a pretest probability of 0.50, a negative FFR-CT would yield a posttest probability of 0.14 (Danad et al [2017]) and 0.12 (Wu et al [2016]). Overall, the negative likelihood ratios and posttest probability estimates for FFR-CT are slightly better than CCTA as well as SECHO and SPECT.
Table 5. Change in Disease Probability Following a Negative Test
 |  |  | Post-test Probability (95% CI) After Negative Test |
| Study | Modality | Negative LR(95% CI) | Pretest Probability 0.25 | Pretest Probability 0.50 | Pretest Probability 0.75 |
| Danad et al (2017)32, |  |  |  |  |  |
 | MRI | 0.12 (0.05 to 0.30) | 0.04 (0.02 to 0.09) | 0.11 (0.05 to 0.23) | 0.26 (0.13 to 0.47) |
 | FFR-CT | 0.16 (0.11 to 0.23) | 0.05 (0.04 to 0.07) | 0.14 (0.10 to 0.19) | 0.32 (0.25 to 0.41) |
 | CCTA | 0.22 (0.10 to 0.50) | 0.07 (0.03 to 0.14) | 0.18 (0.09 to 0.33) | 0.40 (0.23 to 0.60) |
 | SECHO | 0.34 (0.17 to 0.66) | 0.10 (0.05 to 0.18) | 0.25 (0.15 to 0.40) | 0.50 (0.34 to 0.66) |
 | SPECT | 0.40 (0.19 to 0.83) | 0.12 (0.06 to 0.22) | 0.29 (0.16 to 0.45) | 0.55 (0.36 to 0.71) |
 | ICA | 0.46 (0.39 to 0.55) | 0.13 (0.12 to 0.15) | 0.32 (0.28 to 0.35) | 0.58 (0.54 to 0.62) |
| Wu et al (2016)31, |  |  |  |  |  |
 | FFR-CT | 0.14 (0.09 to 0.21) | 0.04 (0.03 to 0.07) | 0.12 (0.08 to 0.17) | 0.30 (0.21 to 0.39) |
| Takx et al (2015)30, |  |  |  |  |  |
 | MRI | 0.14 (0.10 to 0.18) | 0.04 (0.03 to 0.06) | 0.12 (0.09 to 0.15) | 0.30 (0.23 to 0.35) |
 | Perfusion CT | 0.12 (0.04 to 0.33) | 0.04 (0.01 to 0.10) | 0.11 (0.04 to 0.25) | 0.26 (0.11 to 0.50) |
 | SECHO | 0.42 (0.30 to 0.59) | 0.12 (0.09 to 0.16) | 0.30 (0.23 to 0.37) | 0.56 (0.47 to 0.64) |
 | SPECT | 0.39 (0.27 to 0.55) | 0.12 (0.08 to 0.15) | 0.28 (0.21 to 0.35) | 0.54 (0.45 to 0.62) |
 | PET | 0.14 (0.02 to 0.87) | 0.04 (0.01 to 0.22) | 0.12 (0.02 to 0.47) | 0.30 (0.06 to 0.72) |
CCTA: coronary computed tomography angiography; CI: confidence interval; CT: computed tomography; FFR-CT: fractional flow reserve using coronary computed tomography angiography; ICA: invasive coronary angiography; LR: likelihood ratio; MRI: magnetic resonance imaging; PET: positron emission tomography; SECHO: stress echocardiography; SPECT: single-photon emission computed tomography.
Curzen et al (2016) conducted a literature search that identified 1 study that examined 200 consecutive individuals selected from the NXT trial population "to reproduce the methodology of the invasive RIPCORD study" with the elective management of stable chest pain.51, All subjects received CCTA including FFR-CT "in at least 1 vessel with diameter ≥ 2 mm and diameter stenosis ≥ 30%" as well as ICA within 60 days of CCTA. Three experienced interventional cardiologists reviewed the CCTA results (initially without the FFR-CT results) and selected a management plan from the following 4 options: "1) optimal medical therapy (OMT) alone; 2) PCI + OMT; 3) coronary artery bypass graft + OMT; or 4) more information about ischemia required - they committed to option 1 by consensus." Following the initial decision, results from the FFR-CT were shared with the same group of interventional cardiologists who again decided by consensus based on the same 4 options. A cutoff of 0.80 or less was considered significant on FFR-CT. A stenosis was considered significant on CCTA or ICA with 50% or more diameter narrowing. Change in management between the first decision based on CCTA only and the second decision based on CCTA plus FFR-CT was the primary endpoint of this study. Secondary endpoints included analysis of the vessels considered to have significant stenosis based on CCTA alone versus CCTA plus FFR-CT as well as vessels identified as targets for revascularization based on CCTA alone versus CCTA plus FFR-CT. This study was conducted by investigators in the United Kingdom and Denmark. Funding was provided by HeartFlow, and multiple authors reported receiving fees, grants, and/or support from HeartFlow.
Results for the primary endpoint (see Table 6) yielded a change in management category for 72 (36%) of 200 individuals. For the 87 individuals initially assigned to PCI based on CCTA alone, the addition of the FFR-CT results shifted management for 26 (30%) of 87 to OMT (ie, no ischemic lesion on FFR-CT) and an additional 16 (18%) individuals remained in the PCI category but FFR-CT identified a different target vessel for PCI. These findings provide supportive information that the improved diagnostic accuracy of FFR-CT in particular related to its better negative likelihood ratio compared with CCTA alone would likely lead to changes in management that would be expected to improve health outcomes.
Table 6. Summary of Overall Management Changes for Patients Using CCTA Versus CCTA Plus FFR-CT
Management Category
Consensus Decision | CCTA Alone, n (%) | CCTA Plus FFR-CT, n (%) | Strategy Changea (95% CI), % |
| More data required | 38 (19.0) | 0 | NR |
| Optimal medical therapy | 67 (33.5) | 113 (56.5) | 23 (18 to 29) |
| Percutaneous coronary intervention | 87 (43.5) | 78 (39.0) | -5 (-2 to -8) |
| Coronary artery bypass graft surgery | 8 (4.0) | 9 (4.5) | 0.5 (0.1 to 3) |
CCTA: coronary computed tomography angiography; CI: confidence interval; FFR-CT: fractional flow reserve using coronary computed tomography angiography.
a p<.001 for between-group change, CCTA alone versus CCTA + FFR-CT.
Section Summary: Clinically Useful
There is direct evidence, provided by 2 prospective and 2 retrospective studies, that compares health outcomes observed during 90-day to 1-year follow-up for strategies using CCTA particularly in combination with selective FFR-CT with strategies using ICA or other noninvasive imaging tests. The available evidence provides support that use of CCTA with selective FFR-CT is likely to reduce the use of ICA in individuals with stable chest pain who are unlikely to benefit from revascularization by demonstrating the absence of functionally significant obstructive CAD. Also, the benefits are likely to outweigh potential harms given that rates of revascularization for functionally significant obstructive CAD appear to be similar and cardiac-related adverse events do not appear to be increased following a CCTA with selective FFR-CT strategy. Moreover, the evidence on the diagnostic performance characteristics, particularly showing higher specificity of FFR-CT and better negative likelihood ratio as compared with CCTA alone, may be combined with indirect evidence that CCTA with a selective FFR-CT strategy would likely lead to changes in management that would be expected to improve health outcomes, particularly by limiting unnecessary ICA testing. While individual studies are noted to have specific methodologic limitations and some variation is noted in the magnitude of benefit across studies, in aggregate the evidence provides reasonable support that the selective addition of FFR-CT following CCTA results in a meaningful improvement in the net health outcome.
Summary of Evidence
For individuals with stable chest pain at intermediate risk of coronary artery disease (ie, suspected or presumed stable ischemic heart disease) being considered for ICA who receive noninvasive FFR measurement following positive coronary computed tomography angiography (CCTA), the evidence includes both direct and indirect evidence: 2 meta-analyses on diagnostic performance; 1 prospective, multi-center nonrandomized comparative study; 1 prospective cohort; 2 retrospective cohort studies; and 1 study reporting changes in management associated with CCTA-based strategies with selective addition of FFR using CCTA (FFR-CT), and 1 randomized controlled trial comparing of CCTA alone with ICA. Relevant outcomes are test accuracy and validity, morbid events, quality of life, resource utilization, and treatment-related morbidity. The meta-analyses indicated that CCTA has high sensitivity but moderately low specificity for hemodynamically significant obstructive disease. There is direct evidence, provided by 2 prospective and 2 retrospective studies, that compares health outcomes observed during 90-day to 1-year follow-up for strategies using CCTA particularly in combination with selective FFR-CT with strategies using ICA or other noninvasive imaging tests. The available evidence provides support that use of CCTA with selective FFR-CT is likely to reduce the use of ICA in individuals with stable chest pain who are unlikely to benefit from revascularization by demonstrating the absence of functionally significant obstructive coronary artery disease. Also, the benefits are likely to outweigh potential harms because rates of revascularization for functionally significant obstructive coronary artery disease appear to be similar and treatment-related adverse events do not appear to increase following CCTA with a selective FFR-CT strategy. Moreover, given the available evidence that CCTA alone has been used to select patients to avoid ICA, the studies showing higher specificity of FFR-CT and lower negative likelihood ratio of FFR-CT compared with CCTA alone may be used to build a chain of evidence that CCTA with a selective FFR-CT strategy would likely lead to changes in management that would be expected to improve health outcomes by further limiting unnecessary ICA testing. While individual studies are noted to have specific methodologic limitations and some variation has been noted in the magnitude of benefit across studies, in aggregate the evidence provides reasonable support that the selective addition of FFR-CT following CCTA results in a meaningful improvement in the net health outcome. The evidence is sufficient to determine that the technology results in meaningful improvements in the net health outcome.
SUPPLEMENTAL INFORMATION
Practice Guidelines and Position Statements
National Institute for Health and Care Excellence
In 2017, the National Institute for Health and Care Excellence (NICE) endorsed fractional flow reserve using coronary computed tomography angiography (FFR-CT), with the following conclusions: "The committee concluded that the evidence suggests that HeartFlow FFRCT is safe, has high diagnostic accuracy, and that its use may avoid the need for invasive investigations."52,
Recommendations included:
- "The case for adopting HeartFlow FFRCT for estimating fractional flow reserve from coronary CT angiography (CCTA) is supported by the evidence. The technology is non-invasive and safe, and has a high level of diagnostic accuracy."
- "HeartFlow FFRCT should be considered as an option for patients with stable, recent onset chest pain who are offered CCTA as part of the NICE pathway on chest pain. Using HeartFlow FFRCT may avoid the need for invasive coronary angiography and revascularization. For correct use, HeartFlow FFRCT requires access to 64-slice (or above) CCTA facilities."
U.S. Preventive Services Task Force Recommendations
Not applicable.
Ongoing Clinical Trials
Some currently unpublished trials that might influence this policy are listed in Table 7
| NCT No. | Trial Name | Planned Enrollment | Completion Date |
| Ongoing |  |  |  |
| NCT02400229 | Diagnostic Imaging Strategies for Patients With Stable Chest Pain and Intermediate Risk of Coronary Artery Disease: Comparative Effectiveness Research of Existing Technologies) - A Pragmatic Randomised Controlled Trial of CT Versus ICA | 3546 | Apr 2021 |
| NCT02973126 | Assessment of Fractional Flow reservE Computed Tomography Versus Single Photon Emission Computed Tomography in the Diagnosis of Hemodynamically Significant Coronary Artery Disease. (AFFECTS) | 270 | Oct 2020 |
| NCT02208388 | Prospective Evaluation of MyocaRdial PerFUSion ComputEd Tomography Trial | 1000 | Apr 2024 |
| Unpublished |  |  |  |
| NCT02173275 | Computed TomogRaphic Evaluation of Atherosclerotic DEtermiNants of Myocardial IsChEmia (The CREDENCE TRIAL) | 618 | Jan 2018
(updated 11/13/2019) |
NCT: national clinical trial.]
________________________________________________________________________________________
Horizon BCBSNJ Medical Policy Development Process:
This Horizon BCBSNJ Medical Policy (the “Medical Policy”) has been developed by Horizon BCBSNJ’s Medical Policy Committee (the “Committee”) consistent with generally accepted standards of medical practice, and reflects Horizon BCBSNJ’s view of the subject health care services, supplies or procedures, and in what circumstances they are deemed to be medically necessary or experimental/ investigational in nature. This Medical Policy also considers whether and to what degree the subject health care services, supplies or procedures are clinically appropriate, in terms of type, frequency, extent, site and duration and if they are considered effective for the illnesses, injuries or diseases discussed. Where relevant, this Medical Policy considers whether the subject health care services, supplies or procedures are being requested primarily for the convenience of the covered person or the health care provider. It may also consider whether the services, supplies or procedures are more costly than an alternative service or sequence of services, supplies or procedures that are at least as likely to produce equivalent therapeutic or diagnostic results as to the diagnosis or treatment of the relevant illness, injury or disease. In reaching its conclusion regarding what it considers to be the generally accepted standards of medical practice, the Committee reviews and considers the following: all credible scientific evidence published in peer-reviewed medical literature generally recognized by the relevant medical community, physician and health care provider specialty society recommendations, the views of physicians and health care providers practicing in relevant clinical areas (including, but not limited to, the prevailing opinion within the appropriate specialty) and any other relevant factor as determined by applicable State and Federal laws and regulations.
___________________________________________________________________________________________________________________________
Index:
Coronary Computed Tomography Angiography With Selective Noninvasive Fractional Flow Reserve
Noninvasive Fractional Flow Reserve Using Computed Tomography Angiography
Fractional Flow Reserve (FFR), Noninvasive
FFR (Fractional Flow Reserve), Noninvasive
FFRCT
HeartFlow
References:
1. Patel MR, Peterson ED, Dai D, et al. Low diagnostic yield of elective coronary angiography. N Engl J Med. Mar 11 2010; 362(10): 886-95. PMID 20220183
2. Boden WE, O'Rourke RA, Teo KK, et al. Optimal medical therapy with or without PCI for stable coronary disease. N Engl J Med. Apr 12 2007; 356(15): 1503-16. PMID 17387127
3. Fihn SD, Gardin JM, Abrams J, et al. 2012 ACCF/AHA/ACP/AATS/PCNA/SCAI/STS Guideline for the diagnosis and management of patients with stable ischemic heart disease: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, and the American College of Physicians, American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J Am Coll Cardiol. Dec 18 2012; 60(24): e44-e164. PMID 23182125
4. Diamond GA, Forrester JS. Analysis of probability as an aid in the clinical diagnosis of coronary-artery disease. N Engl J Med. Jun 14 1979; 300(24): 1350-8. PMID 440357
5. Genders TS, Steyerberg EW, Alkadhi H, et al. A clinical prediction rule for the diagnosis of coronary artery disease: validation, updating, and extension. Eur Heart J. Jun 2011; 32(11): 1316-30. PMID 21367834
6. Wasfy MM, Brady TJ, Abbara S, et al. Comparison of the Diamond-Forrester method and Duke Clinical Score to predict obstructive coronary artery disease by computed tomographic angiography. Am J Cardiol. Apr 01 2012; 109(7): 998-1004. PMID 22236462
7. Versteylen MO, Joosen IA, Shaw LJ, et al. Comparison of Framingham, PROCAM, SCORE, and Diamond Forrester to predict coronary atherosclerosis and cardiovascular events. J Nucl Cardiol. Oct 2011; 18(5): 904-11. PMID 21769703
8. Min JK, Dunning A, Gransar H, et al. Medical history for prognostic risk assessment and diagnosis of stable patients with suspected coronary artery disease. Am J Med. Aug 2015; 128(8): 871-8. PMID 25865923
9. Genders TS, Steyerberg EW, Hunink MG, et al. Prediction model to estimate presence of coronary artery disease: retrospective pooled analysis of existing cohorts. BMJ. Jun 12 2012; 344: e3485. PMID 22692650
10. CAD Consortium. Pre-test probability of CAD. 2016; https://www.qxmd.com/calculate/calculator_287/pre-test- probability-of-cad-cad-consortium. Accessed March 19, 2020.
11. De Bruyne B, Fearon WF, Pijls NH, et al. Fractional flow reserve-guided PCI for stable coronary artery disease. N Engl J Med. Sep 25 2014; 371(13): 1208-17. PMID 25176289
12. De Bruyne B, Pijls NH, Kalesan B, et al. Fractional flow reserve-guided PCI versus medical therapy in stable coronary disease. N Engl J Med. Sep 13 2012; 367(11): 991-1001. PMID 22924638
13. Tonino PA, De Bruyne B, Pijls NH, et al. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention. N Engl J Med. Jan 15 2009; 360(3): 213-24. PMID 19144937
14. Pothineni NV, Shah NN, Rochlani Y, et al. U.S. Trends in Inpatient Utilization of Fractional Flow Reserve and Percutaneous Coronary Intervention. J Am Coll Cardiol. Feb 16 2016; 67(6): 732-733. PMID 26868697
15. Blue Cross Blue Shield Association Technology Evaluation Center (TEC). Fractional Flow Reserve and Coronary Artery Revascularization. TEC Assessment. June 2011;26:Tab 2.
16. Fearon WF, Shilane D, Pijls NH, et al. Cost-effectiveness of percutaneous coronary intervention in patients with stable coronary artery disease and abnormal fractional flow reserve. Circulation. Sep 17 2013; 128(12): 1335-40. PMID 23946263
17. van Nunen LX, Zimmermann FM, Tonino PA, et al. Fractional flow reserve versus angiography for guidance of PCI in patients with multivessel coronary artery disease (FAME): 5-year follow-up of a randomised controlled trial. Lancet. Nov 07 2015; 386(10006): 1853-60. PMID 26333474
18. Montalescot G, Sechtem U, Achenbach S, et al. 2013 ESC guidelines on the management of stable coronary artery disease: the Task Force on the management of stable coronary artery disease of the European Society of Cardiology. Eur Heart J. Oct 2013; 34(38): 2949-3003. PMID 23996286
19. Windecker S, Kolh P, Alfonso F, et al. 2014 ESC/EACTS Guidelines on myocardial revascularization: The Task Force on Myocardial Revascularization of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS)Developed with the special contribution of the European Association of Percutaneous Cardiovascular Interventions (EAPCI). Eur Heart J. Oct 01 2014; 35(37): 2541-619. PMID 25173339
20. Patel MR, Calhoon JH, Dehmer GJ, et al. ACC/AATS/AHA/ASE/ASNC/SCAI/SCCT/STS 2017 Appropriate Use Criteria for Coronary Revascularization in Patients With Stable Ischemic Heart Disease: A Report of the American College of Cardiology Appropriate Use Criteria Task Force, American Association for Thoracic Surgery, American Heart Association, American Society of Echocardiography, American Society of Nuclear Cardiology, Society for Cardiovascular Angiography and Interventions, Society of Cardiovascular Computed Tomography, and Society of Thoracic Surgeons. J Am Coll Cardiol. May 02 2017; 69(17): 2212-2241. PMID 28291663
21. Pijls NH, Van Gelder B, Van der Voort P, et al. Fractional flow reserve. A useful index to evaluate the influence of an epicardial coronary stenosis on myocardial blood flow. Circulation. Dec 01 1995; 92(11): 3183-93. PMID 7586302
22. de Bruyne B, Bartunek J, Sys SU, et al. Simultaneous coronary pressure and flow velocity measurements in humans. Feasibility, reproducibility, and hemodynamic dependence of coronary flow velocity reserve, hyperemic flow versus pressure slope index, and fractional flow reserve. Circulation. Oct 15 1996; 94(8): 1842-9. PMID 8873658
23. HeartFlow. De Novo Classification Request for FFRct v. 1.4. 2013; https://www.accessdata.fda.gov/cdrh_docs/reviews/DEN130045.pdf. Accessed March 19, 2020.
24. Koo BK, Erglis A, Doh JH, et al. Diagnosis of ischemia-causing coronary stenoses by noninvasive fractional flow reserve computed from coronary computed tomographic angiograms. Results from the prospective multicenter DISCOVER-FLOW (Diagnosis of Ischemia-Causing Stenoses Obtained Via Noninvasive Fractional Flow Reserve) study. J Am Coll Cardiol. Nov 01 2011; 58(19): 1989-97. PMID 22032711
25. Min JK, Koo BK, Erglis A, et al. Effect of image quality on diagnostic accuracy of noninvasive fractional flow reserve: results from the prospective multicenter international DISCOVER-FLOW study. J Cardiovasc Comput Tomogr. May-Jun 2012; 6(3): 191-9. PMID 22682261
26. Nakazato R, Park HB, Berman DS, et al. Noninvasive fractional flow reserve derived from computed tomography angiography for coronary lesions of intermediate stenosis severity: results from the DeFACTO study. Circ Cardiovasc Imaging. Nov 2013; 6(6): 881-9. PMID 24081777
27. Norgaard BL, Leipsic J, Gaur S, et al. Diagnostic performance of noninvasive fractional flow reserve derived from coronary computed tomography angiography in suspected coronary artery disease: the NXT trial (Analysis of Coronary Blood Flow Using CT Angiography: Next Steps). J Am Coll Cardiol. Apr 01 2014; 63(12): 1145-1155. PMID 24486266
28. Taylor CA, Fonte TA, Min JK. Computational fluid dynamics applied to cardiac computed tomography for noninvasive quantification of fractional flow reserve: scientific basis. J Am Coll Cardiol. Jun 04 2013; 61(22): 2233-41. PMID 23562923
29. Kim KH, Doh JH, Koo BK, et al. A novel noninvasive technology for treatment planning using virtual coronary stenting and computed tomography-derived computed fractional flow reserve. JACC Cardiovasc Interv. Jan 2014; 7(1): 72-8. PMID 24332418
30. Takx RA, Blomberg BA, El Aidi H, et al. Diagnostic accuracy of stress myocardial perfusion imaging compared to invasive coronary angiography with fractional flow reserve meta-analysis. Circ Cardiovasc Imaging. Jan 2015; 8(1). PMID 25596143
31. Wu W, Pan DR, Foin N, et al. Noninvasive fractional flow reserve derived from coronary computed tomography angiography for identification of ischemic lesions: a systematic review and meta-analysis. Sci Rep. Jul 05 2016; 6: 29409. PMID 27377422
32. Danad I, Szymonifka J, Twisk JWR, et al. Diagnostic performance of cardiac imaging methods to diagnose ischaemia-causing coronary artery disease when directly compared with fractional flow reserve as a reference standard: a meta-analysis. Eur Heart J. Apr 01 2017; 38(13): 991-998. PMID 27141095
33. Renker M, Schoepf UJ, Wang R, et al. Comparison of diagnostic value of a novel noninvasive coronary computed tomography angiography method versus standard coronary angiography for assessing fractional flow reserve. Am J Cardiol. Nov 01 2014; 114(9): 1303-8. PMID 25205628
34. De Geer J, Sandstedt M, Bjorkholm A, et al. Software-based on-site estimation of fractional flow reserve using standard coronary CT angiography data. Acta Radiol. Oct 2016; 57(10): 1186-92. PMID 26691914
35. Wardziak L, Kruk M, Pleban W, et al. Coronary CTA enhanced with CTA based FFR analysis provides higher diagnostic value than invasive coronary angiography in patients with intermediate coronary stenosis. J Cardiovasc Comput Tomogr. Jan 2019; 13(1): 62-67. PMID 30309764
36. Min JK, Leipsic J, Pencina MJ, et al. Diagnostic accuracy of fractional flow reserve from anatomic CT angiography. JAMA. Sep 26 2012; 308(12): 1237-45. PMID 22922562
37. Thompson AG, Raju R, Blanke P, et al. Diagnostic accuracy and discrimination of ischemia by fractional flow reserve CT using a clinical use rule: results from the Determination of Fractional Flow Reserve by Anatomic Computed Tomographic Angiography study. J Cardiovasc Comput Tomogr. Mar-Apr 2015; 9(2): 120-8. PMID 25819194
38. Whiting PF, Rutjes AW, Westwood ME, et al. QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med. Oct 18 2011; 155(8): 529-36. PMID 22007046
39. Coenen A, Lubbers MM, Kurata A, et al. Fractional flow reserve computed from noninvasive CT angiography data: diagnostic performance of an on-site clinician-operated computational fluid dynamics algorithm. Radiology. Mar 2015; 274(3): 674-83. PMID 25322342
40. PROSPERO. International prospective register of systematic reviews. n.d.; https://www.crd.york.ac.uk/PROSPERO/. Accessed March 19, 2020.
41. Min JK, Berman DS, Budoff MJ, et al. Rationale and design of the DeFACTO (Determination of Fractional Flow Reserve by Anatomic Computed Tomographic AngiOgraphy) study. J Cardiovasc Comput Tomogr. Sep-Oct 2011; 5(5): 301-9. PMID 21930103
42. Douglas PS, De Bruyne B, Pontone G, et al. 1-Year Outcomes of FFRCT-Guided Care in Patients With Suspected Coronary Disease: The PLATFORM Study. J Am Coll Cardiol. Aug 02 2016; 68(5): 435-445. PMID 27470449
43. Douglas PS, Pontone G, Hlatky MA, et al. Clinical outcomes of fractional flow reserve by computed tomographic angiography-guided diagnostic strategies vs. usual care in patients with suspected coronary artery disease: the prospective longitudinal trial of FFR(CT): outcome and resource impacts study. Eur Heart J. Dec 14 2015; 36(47): 3359-67. PMID 26330417
44. Hlatky MA, De Bruyne B, Pontone G, et al. Quality-of-Life and Economic Outcomes of Assessing Fractional Flow Reserve With Computed Tomography Angiography: PLATFORM. J Am Coll Cardiol. Dec 01 2015; 66(21): 2315-2323. PMID 26475205
45. Dewey M, Rief M, Martus P, et al. Evaluation of computed tomography in patients with atypical angina or chest pain clinically referred for invasive coronary angiography: randomised controlled trial. BMJ. Oct 24 2016; 355: i5441. PMID 27777234
46. Jensen JM, Botker HE, Mathiassen ON, et al. Computed tomography derived fractional flow reserve testing in stable patients with typical angina pectoris: influence on downstream rate of invasive coronary angiography. Eur Heart J Cardiovasc Imaging. Apr 01 2018; 19(4): 405-414. PMID 28444153
47. Norgaard BL, Hjort J, Gaur S, et al. Clinical Use of Coronary CTA-Derived FFR for Decision-Making in Stable CAD. JACC Cardiovasc Imaging. May 2017; 10(5): 541-550. PMID 27085447
48. Lu MT, Ferencik M, Roberts RS, et al. Noninvasive FFR Derived From Coronary CT Angiography: Management and Outcomes in the PROMISE Trial. JACC Cardiovasc Imaging. Nov 2017; 10(11): 1350-1358. PMID 28412436
49. Douglas PS, Hoffmann U, Lee KL, et al. PROspective Multicenter Imaging Study for Evaluation of chest pain: rationale and design of the PROMISE trial. Am Heart J. Jun 2014; 167(6): 796-803.e1. PMID 24890527
50. Patel MR, Norgaard BL, Fairbairn TA, et al. 1-Year Impact on Medical Practice and Clinical Outcomes of FFR CT : The ADVANCE Registry. JACC Cardiovasc Imaging. Jan 2020; 13(1 Pt 1): 97-105. PMID 31005540
51. Curzen NP, Nolan J, Zaman AG, et al. Does the Routine Availability of CT-Derived FFR Influence Management of Patients With Stable Chest Pain Compared to CT Angiography Alone?: The FFR CT RIPCORD Study. JACC Cardiovasc Imaging. Oct 2016; 9(10): 1188-1194. PMID 27568119
52. National Institute for Health and Care Excellence. HeartFlow FFRCT for estimating fractional flow reserve from coronary CT angiography [MTG32]. 2017; https://www.nice.org.uk/guidance/mtg32. Accessed March 19, 2020.
Codes:
(The list of codes is not intended to be all-inclusive and is included below for informational purposes only. Inclusion or exclusion of a procedure, diagnosis, drug or device code(s) does not constitute or imply authorization, certification, approval, offer of coverage or guarantee of payment.)
CPT*
0501T
0502T
0503T
0504T
HCPCS
* CPT only copyright 2020 American Medical Association. All rights reserved. CPT is a registered trademark of the American Medical Association.
_________________________________________________________________________________________
Medical policies can be highly technical and are designed for use by the Horizon BCBSNJ professional staff in making coverage determinations. Members referring to this policy should discuss it with their treating physician, and should refer to their specific benefit plan for the terms, conditions, limitations and exclusions of their coverage.
The Horizon BCBSNJ Medical Policy Manual is proprietary. It is to be used only as authorized by Horizon BCBSNJ and its affiliates. The contents of this Medical Policy are not to be copied, reproduced or circulated to other parties without the express written consent of Horizon BCBSNJ. The contents of this Medical Policy may be updated or changed without notice, unless otherwise required by law and/or regulation. However, benefit determinations are made in the context of medical policies existing at the time of the decision and are not subject to later revision as the result of a change in medical policy
____________________________________________________________________________________________________________________________ |